 eCOO® Technology with scCO2 sterilisation is Leader Biomedical’s unique proprietary solution utilising scCO2 to clean, sterilise, and impregnate human bone, animal-derived bone, soft tissue and synthetic grafts.
eCOO® Technology with scCO2 sterilisation is Leader Biomedical’s unique proprietary solution utilising scCO2 to clean, sterilise, and impregnate human bone, animal-derived bone, soft tissue and synthetic grafts.
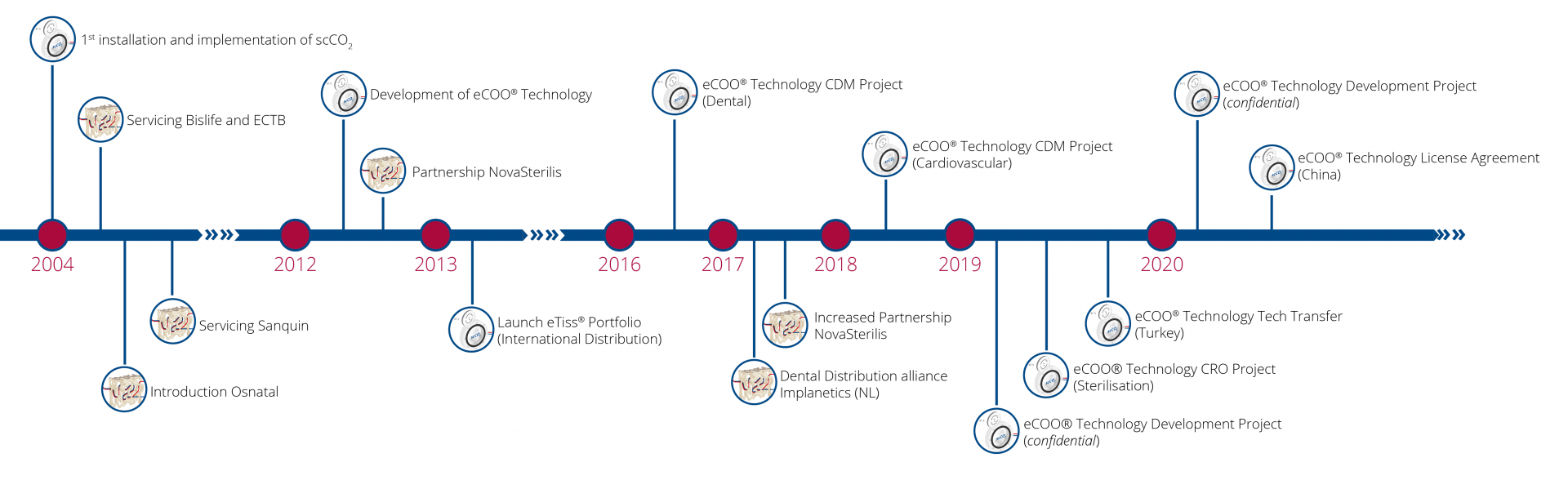
The eCOO® platform has built experience since 2004 on processing for medical applications and distributing products worldwide. This has allowed us to gain insight in different markets and develop various promotional strategies.
eCOO® Technology

eCOO® Technology utilises a breakthrough solution based on supercritical carbon dioxide (scCO2). Under a certain pressure and temperature, carbon dioxide enters a supercritical state in which it exhibits both the properties of a gas and a liquid.
eCOO® Technology is ideal for cleaning (e.g. decellularising) and sterilising tissue, because it can permeate matter like a gas and dissolve and cleanse it like a liquid at macro- and micropore level. By using scCO2 as the cleaning solvent, commonly used processing with excessive incubation in detergents becomes unnecessary.
✓ reducing risk of processing residuals
✓ retaining the extra-cellular matrix (ECM)
Leader Biomedical has successfully applied eCOO® Technology to process human-derived cancellous and cortical bone, and tendons for over 10 years in Europe. eCOO® Technology has also been used to develop a dental membrane, derived from porcine tissue, for applications in guided bone and tissue regeneration. eCOO® Technology has further been applied by partners in various projects and used in a number of clinical studies.
What our customers say:
“scCO2 treatment is the ideal first step of any cascade of tissue treatment, since it almost completely eliminates the lipid fraction of tissues without toxic solvents”
eCOO® Technology Platform
our expertise
eCOO® exclusively focuses on supporting the designers of non-cellularised products or components. By utilising specific properties of scCO2 to address sensitive materials for commercial applications, we offer scalable technology that can create safe and compliant novel products.
our experience
The eCOO® Technology network has access to partners capable of manufacturing and advising on a product that complies to our client’s regulatory strategy, such as CE marking, FDA or other.

 Global
Global  Nederland
Nederland APAC
APAC India
India